Hydrogen Fuel Saver - Technical and Safety Data
You may have heard all sorts of stories (from people who haven't done their research properly!) about how these HHO systems don't work or are dangerous. Well that's absolute rubbish!
We have LOTS of scientific data below for those techies amongst you. Take a look at the video below first to find out EXACTLY how a hydrogen on demand system works, and then read the scientific studies below that:

NASA conducted several studies (CLICK HERE TO SEE THE FULL PDF!) using hydrogen as a supplemental fuel on a internal combustion engine running on gasoline. Their research specifically demonstrated that the higher flame speed of hydrogen was responsible for being able to extend the efficient lean operating range of a gasoline engine. Lean-mixture-ratio combustion in internal-combustion engines has the potential of producing low emissions and higher thermal efficiency for several reasons:
1. Excess oxygen in the charge further oxidizes unburned hydrocarbons and carbon monoxide;
2. Excess oxygen lowers the peak combustion temperatures, which inhibits the formation of oxides of nitrogen;
3. The lower combustion temperatures increase the mixture specific heat ratio by decreasing the net dissociation losses;
4. As the specific heat ratio increases, the cycle thermal efficiency also increases, which gives the potential for better fuel economy.
 CALTECH - California Institute of Technology also made some research (CLICK HERE FOR THE FULL PDF) calculating the thermal efficiency on system fuel economy using supplemental hydrogen. The overall engine efficiency increases and is more dominant than the energy loss incurred in generating hydrogen, resulting improved fuel economy for the system as a whole.
CALTECH - California Institute of Technology also made some research (CLICK HERE FOR THE FULL PDF) calculating the thermal efficiency on system fuel economy using supplemental hydrogen. The overall engine efficiency increases and is more dominant than the energy loss incurred in generating hydrogen, resulting improved fuel economy for the system as a whole.
 Alexandria University - (CLICK HERE FOR THE FULL STUDY REPORT) The objective of this work was to construct a simple innovative HHO generation system and evaluate the effect of hydroxyl gas HHO addition, as an engine performance improver, into gasoline fuel on engine performance and emissions. HHO cell was designed, fabricated and optimized for maximum HHO gas productivity per input power. The optimized parameters were the number of neutral plates, distance between them and type and quantity of two catalysts of Potassium Hydroxide (KOH) and sodium hydroxide (NaOH). The performance of a Skoda Felicia 1.3 GLXi gasoline engine was evaluated with and without the optimized HHO cell. In addition, the CO, HC and NOx emissions were measured using TECNO TEST exhaust gas analyser TE488. The results showed that the HHO gas maximum productivity of the cell was 18 L/h when using 2 neutrals plates with 1 mm distance and 6 g/L of KOH. The results also showed 10% increment in the gasoline engine thermal efficiency, 34% reduction in fuel consumption, 18% reduction in CO, 14% reduction in HC and 15% reduction in NOx.
Alexandria University - (CLICK HERE FOR THE FULL STUDY REPORT) The objective of this work was to construct a simple innovative HHO generation system and evaluate the effect of hydroxyl gas HHO addition, as an engine performance improver, into gasoline fuel on engine performance and emissions. HHO cell was designed, fabricated and optimized for maximum HHO gas productivity per input power. The optimized parameters were the number of neutral plates, distance between them and type and quantity of two catalysts of Potassium Hydroxide (KOH) and sodium hydroxide (NaOH). The performance of a Skoda Felicia 1.3 GLXi gasoline engine was evaluated with and without the optimized HHO cell. In addition, the CO, HC and NOx emissions were measured using TECNO TEST exhaust gas analyser TE488. The results showed that the HHO gas maximum productivity of the cell was 18 L/h when using 2 neutrals plates with 1 mm distance and 6 g/L of KOH. The results also showed 10% increment in the gasoline engine thermal efficiency, 34% reduction in fuel consumption, 18% reduction in CO, 14% reduction in HC and 15% reduction in NOx.
 Engine´s Torque
Engine´s Torque
An average of 19.1% increment in engine torque is obtained using HHO compared to pure carbon fuel operation (e.g. diesel in the graph below). The increase in power is due to the oxygen concentration of HHO gas and to a better mixing of HHO with air and fuel that yields enhanced combustion.
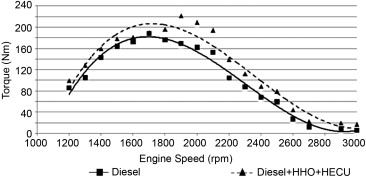
The results show that the addition of HHO can significantly enlarge the flammable region and extend the flammability limit to lower equivalence ratios. Since HHO gas has a low ignition energy and fast flame speed, the HHO and diesel mixture can be more easily ignited and more quickly combusted than the pure diesel fuel. The result is that improved torques at high speeds can be obtained.
 Engine´s Temperature
Engine´s Temperature
High laminar flame velocity of HHO yields decreased ignition delay and shortened combustion period providing lower heat losses, much closer to ideal constant-volume combustion which results in increased compression ratio and thermal efficiency. The result is lower engine temperatures.
 Engine´s Noise
Engine´s Noise
High burning velocity of hydroxy provides faster increment in pressure and temperature which may minimize the knocking especially at idle conditions (low or no load). The result is that diminished engine noise.
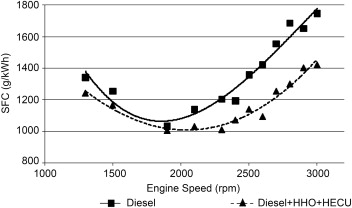
 Fuel Consumption
Fuel Consumption
An average gain of more than 25% is achieved on SFC (Specific Fuel Consumption) by using a HHO System. The reduction in fuel consumption is due to uniform mixing of HHO with air (high diffusivity of HHO) as the higher presence of oxygen which assists diesel during combustion process and yields better combustion. The higher savings occur at high speeds because diesel fuel is hard to be completely burnt at lean conditions due to the increased residual gas fraction and poor mixing. Since HHO gains a high flame speed and wide flammability, the addition of hydrogen will help the fuel to be burned faster and more complete.
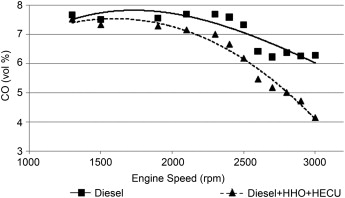
 Carbon Emissions
Carbon Emissions
An average reduction of 35% is gained in CO emissions at mid and high engine speeds. The absence of carbon in HHO gas is a major reason for CO reduction. The HHO and diesel fuel mixture burns faster and more completely than the pure diesel fuel (as in the graph below). Thus, CO emission at high speed and lean conditions is effectively reduced after HHO addition. Since HHO gas contains oxygen, higher combustion efficiency is obtained. The result is that CO emissions are lower.
FUEL SAVER CALCULATOR
